Highlights From the Mass General Brigham 2025 Gene and Cell Therapy Institute Research Symposium
By Steve Ouellette
Overview
The Mass General Brigham 2025 Gene and Cell Therapy Institute Research Symposium took place December 11-12, 2025, featuring presentations from Boston/Cambridge scientists and keynotes from Dr. Carl June and Dr. David Liu. The event balanced academic rigor with commercial development focus, with speakers actively engaged in startups and industry partnerships.
A notable highlight included remarks from former FDA Center for Biologic Evaluation Director Peter Marks, who participated in a fireside chat with Mimi Lee discussing regulatory developments.
Key Themes
Four major trends emerged across presentations:
Technology vs. Translation Barriers: "The core technologies work. The limiting factors are manufacturing, regulatory repetition, patient stratification, and the economics" of developing therapies for rare genetic diseases. Basic science no longer represents the primary constraint.
Platform Strategy Dominance: Speakers consistently emphasized platform approaches that distribute development risk across multiple patients, mutations, or indications — whether through engineered CAR-T cells, mutation-agnostic gene integration, or disease-agnostic editing strategies.
Precision Enabling Scalability: Advanced engineering in gene editors, delivery systems, and computational design support faster timelines and safer profiles while opening new regulatory pathways.
Manufacturing as Critical Enabler and Constraint: Progress includes shortened vein-to-vein times for autologous CAR-T (from 22+ days to ~3 days, targeting ~1 day), automation, closed systems, and in vivo LNP delivery. Persistent gaps include reproducibility at scale, cost, regulatory requirements around Chemistry Manufacturing Controls (CMC), and lack of standardized reusable platforms.
Dr. Carl June's Keynote
CAR-T Evolution and Current State
Dr. June traced CAR-T development from 1980s receptor-fusion concepts through Penn's 2010 clinical work. The field now represents a mature global industry with seven FDA-approved autologous products treating approximately 50,000 patients worldwide.
Manufacturing advancement stands out particularly: vein-to-vein processing time has compressed dramatically. "Shrinking vein-to-vein time, from historically ~22+ days to currently running trials with ~3 days, and expecting to reach ~1 day soon" represents significant progress toward accessibility.
Next-Generation Approaches
Armored CAR-T Cells: An investigator-initiated trial in CAR-refractory lymphoma engineered CD19 CAR-T cells to secrete IL-18 with favorable safety mechanisms. Results showed approximately 80% response rate among 21 heavily pretreated patients, with unexpected potency at low doses. This validates engineered cytokine payloads can reshape microenvironments safely.
Receptor Rewiring: Engineering CAR-T cells expressing IL-9 receptors (normally absent on T cells) produced unexpected improvements with repeated antigen exposure. Data showed STAT4 upregulation and reduced stress response signaling in solid-tumor mouse models.
Solid Tumor Strategy: Rather than purely target-focused approaches, solid tumors require addressing the immunosuppressive tumor microenvironment — hypoxia, acidity, fibroblast barriers. Solutions combine CAR-T intrinsic upgrades plus external combination therapies remodeling the microenvironment.
Multiplex Editing and In Vivo Engineering
Base editing overcomes limitations with multiple Cas9 edits that create problematic double-strand breaks. "Base editing with an ABE enabled ~20 simultaneous edits at high efficiency, opening design space for allogeneic/off-the-shelf products."
In vivo CAR therapies using lipid nanoparticles represent the next frontier, eliminating complex manufacturing while enabling direct T-cell reprogramming via simple injection. This approach particularly suits autoimmune indications, with positive data in systemic lupus erythematosus, idiopathic myopathy, scleroderma, and fibrosis. A German study reported 100% response rates using existing CAR-T therapy for immune system reset.
Regulatory Landscape Fireside Chat
Panelists: Peter Marks, MD, PhD (Former FDA Director) and Mimi Lee, MD, PhD Moderator: Angela Shen, MD, MBA
Navigating 2025 Regulatory Environment
For preclinical and early-stage programs, regulatory turbulence likely resolves before filing. "For Phase 3+ assets, don't assume the U.S. is the only path. Other regulators (UK, Australia, China, etc.) are 'open for business.'" This approach transforms instability into opportunity for testing innovative regulatory concepts.
Platform Regulatory Paradigm
The current system over-penalizes false positives while accepting false negatives in tiny populations. For ultra-rare genetic diseases, requiring bespoke animal models and repeated toxicology studies becomes economically irrational. A more pragmatic approach would "approve bounded platforms (e.g., a defined editor + delivery system + manufacturing controls) so developers can move between constructs without restarting from zero."
Manufacturing as Primary Constraint
Manufacturing reproducibility and characterization at scale represents the most common Complete Response Letter driver, particularly for AAV vectors. Solutions require automation and standardized micro-factories — increasingly feasible as modalities shift toward LNP/mRNA/editors behaving more like chemistry than biology. "Plan earlier and automate aggressively."
Sequencing Infrastructure and Data Governance
Universal sequencing accelerates diagnosis, cohort identification, and therapy design. However, implementation requires balancing centralized infrastructure with patient-controlled data and privacy protections. The U.S. lags other countries partly due to distrust of centralized data and state-level newborn screening variation. Solutions require opt-out provisions and patient education.
Distinguishing Regulatory Substance from Opinion
"Much of what the FDA has been saying publicly through issued guidance and editorials are merely opinion pieces and either aren't substantive or will be challenged in court." Examples include superiority trial implications and Pediatric Priority Review Voucher expiration affecting rare disease funding. Professional legal guidance helps differentiate rhetorical noise from consequential policy shifts.
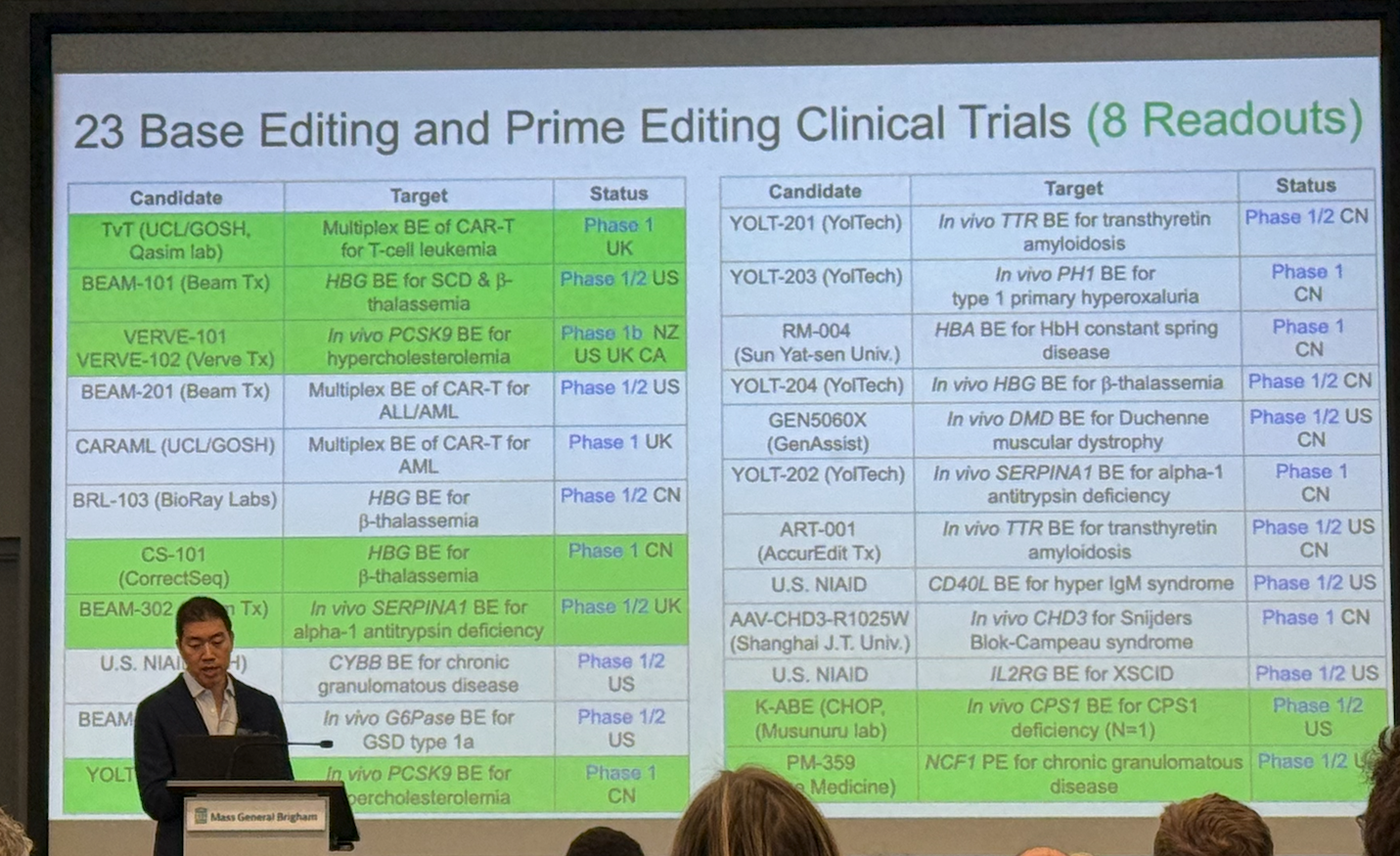
Dr. David Liu's Keynote
Three Editing Paradigms
Dr. Liu organized therapeutic genome editing into three categories mapping to distinct commercialization strategies:
- Mutation-specific correction: Base and prime editing targeting individual mutations
- Mutation-agnostic repair: Targeted gene integration addressing any allele
- Disease-agnostic rescue: Pathway-level fixes unifying patients across genes
Scientific design directly addresses non-scientific bottlenecks including regulatory repetition, manufacturing costs, and population fragmentation.
Clinical Successes in 2025
"Base and prime editors, engineered to avoid double-strand breaks, are now in 20+ clinical trials (mostly in vivo), with eight reporting patient benefit." Notable achievements include:
- Durable remission from base-edited CAR-T in T-cell leukemia
- Autologous HSC base editing for sickle cell disease eliminating pain crises
- In vivo PCSK9 base editing reducing LDL by ~60%
- First direct pathogenic mutation correction in human for alpha-1 antitrypsin deficiency
- Single-patient CPS1 deficiency editor developed in ~7 months
- Prime editing showing early efficacy in chronic granulomatous disease
Mutation-Agnostic Repair Platforms
Two routes preserve native gene regulation:
PASSIGE (prime editing + recombinase landing sites) enables sequential edits after initial insertion.
EvoCAST (evolved CRISPR-associated transposases) boosted mammalian integration 200-350x over natural variants, achieving 10-30% targeted insertion at disease loci without indels — a practical threshold for loss-of-function disorders.
Disease-Agnostic Editing
PERT (prime-editing endogenous tRNAs) rescues premature stop codons affecting approximately 25-30% of pathogenic alleles using a single therapeutic composition. Testing across Batten, Tay-Sachs, and Niemann-Pick diseases restored 20-70% function with minimal global translation disruption. In vivo Hurler syndrome rescue demonstrates scalability for treating many rare diseases without one-drug-per-mutation economics.
Dr. Ben Kleinstiver's Presentation
PAM-Constraint Solutions
Wild-type Cas9 binding requires compatible PAM sequences, blocking many clinically relevant mutations. Dr. Kleinstiver's group developed PAM-relaxed and near-PAMless enzymes accessing nearly any base. However, broader genome access increases off-target encounter risk since PAMs normally provide targeting specificity.
Machine-Learning-Designed Editors
Rather than relying on single PAMless editors, a graduate student, Rachel Silverstein, characterized approximately 1,000 engineered Cas9 variants and trained machine-learning models mapping sequence to targeting preferences. The practical workflow accepts target sequences (with optional off-target avoidance inputs) and outputs custom Cas9 protein sequences predicted to maximize on-target activity while minimizing undesired binding — enabling single-nucleotide allele discrimination.
Clinical Translation: Baby KJ Case
An infant with CPS1 urea cycle disorder had a base-editable mutation but wild-type Cas9 lacked compatible PAMs. A PAMless editor worked but raised safety concerns. An ML-designed Cas9 delivered near-saturating editing with enhanced selectivity, reducing off-target risk. The approach reduced ammonia levels and enabled discharge home on a near-normal diet.
Large DNA Insertion and Immune Evasion
Recombinases and transposases require large dsDNA donors, but cytosolic dsDNA triggers cGAS-STING-mediated toxicity. Dr. Kleinstiver's lab developed immune-evasive "chimeric" donors — circular ssDNA with short dsDNA patches where enzymes bind. LNP-packaged recombinases with chimeric donors administered in vivo tolerated multi-mg/kg dosing with minimal STING activation, achieving ~1% bulk liver integration and potentially enabling non-viral in vivo insertion for select loss-of-function diseases.
Need a custom competitive, IP, or diligence review for your team?
Solidus Bio delivers analyst-grade research for biotech companies and investors making high-stakes decisions.
Book a Scoping Call